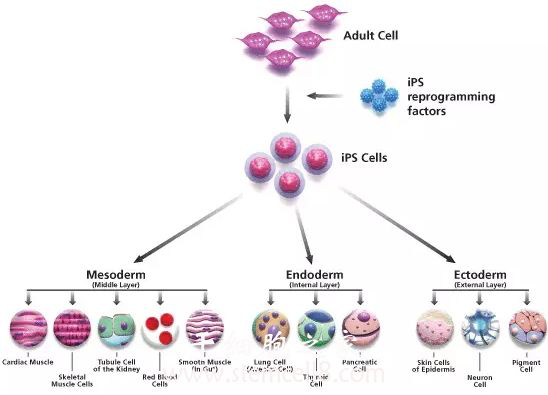
(G) Chromatin immuno-precipitation assays comparing the levels of histone H3 methylation at K4 (H3K4me2), K27 (H3K27me3) and K9 (H3K9me3) in the promoters of OCT4, NANOG, HOXB4 and HOXB5 in human fibroblasts and CD133+ cells. (E) Specific in vitro differentiation of CBiPS2F-1 and (F) CBiPS3F-12 into dopaminergic neurons (Tuj1/TH tyrosine hydroxilase), which are immunophenotypically mature. (D) Immunofluorescence analysis of teratoma sections 60 days after intra-testicular injection of CBiPS2F-1 showing Tuj1/GFAP positive ectoderm, AFP/FoxA2 positive endoderm and ASM/ASA positive mesoderm. (C) In vitro differentiation of CBiPS 2F-1 into the three primary germ cell layers (Ectoderm-Tuj1, Endoderm-AFP and FOXA2, and Mesoderm-ASA and GATA4). (B) Quantitative RT-PCR showing the repression of the OCT4, SOX2, KLF4 and c-MYC transgenes in the CBiPS cell lines. ES and Keratinocyte-iPS (KiPS) cell lines were analysed together with the different CBiPS cell lines derived from fresh and frozen samples. Abbreviations: BMP4, bone morphogenetic protein 4 D, day EB, erythroid body EGF2, epidermal growth factor 2 FGF-2, fibroblast growth factor 2 HPCs, hematopoietic progenitor cells IL, interleukin MK, megakaryocyte SCF, stem cell factor TPO, thrombopoietin.(A) Quantitative RT-PCR analysis for pluripotency markers OCT4, SOX2, NANOG, REX1, CRIPTO, KLF4 and c-MYC. Conventional embryonic stem cells (ESCs) or induced pluripotent stem cells (iPSCs) derived from primates resemble mouse epiblast stem cells, raising an intriguing question regarding whether the naive pluripotent state resembling mouse embryonic stem cells (mESCs) exists in primates and how to captur. (D): The cell number increment of suspension cells from day 14 to day 19 in one well of a 6-well plate. The yellow arrows indicate some of the morphologically larger cells on day 19. (C): Phase contrast images of suspension cells on days 14 and 19. Ig isotype controls for CD41, CD42a, CD34, and CD45 were used to determine the background. The suspension cells expressed MK markers CD41 and CD42a and hematopoietic cell markers CD34 and CD45. (B): Representative flow cytometry dot plots of suspension cells on day 14 (left) and day 19 (right). (A): Schematic diagram for MK generation and expression of CD34, CD45, CD41, and CD42a during hiPSC differentiation. Generation of MKs derived from human induced pluripotent stem cells (hiPSCs). Human induced pluripotent stem cells Megakaryocytes Plasbumin Romiplostim Thrombopoietin. Additional maturation and investigation of hiPSC-derived MKs should provide insights into MK biology and lead to the generation of large numbers of platelets ex vivo. Transcriptome analysis by RNA sequencing revealed that megakaryocytic-related genes were highly expressed. After 19 days, we observed abundant CD41(+)CD42a(+) MKs that also expressed the MK markers CD42b and CD61 and displayed polyploidy (≥16% of derived cells with DNA contents >4N). We used this method to induce MK generation from hiPSCs derived from 23 individuals in two steps: generation of CD34(+)CD45(+) hematopoietic progenitor cells (HPCs) for 14 days and generation and expansion of CD41(+)CD42a(+) MKs from HPCs for an additional 5 days. Several crucial reagents were evaluated and replaced with Food and Drug Administration-approved pharmacological reagents, including romiplostim (Nplate, a thrombopoietin analog), oprelvekin (recombinant interleukin-11), and Plasbumin (human albumin). In this study, we developed an efficient system to generate MKs from hiPSCs under a feeder-free and xeno-free condition, in which all animal-derived products were eliminated. The key requirements in this approach include developing a robust and consistent method to produce functional progeny cells, such as MKs from hiPSCs, and minimizing the risk and variation from the animal-derived products in cell cultures. Ex vivo generation of MKs from human induced pluripotent stem cells (hiPSCs) provides a renewable cell source of platelets for treating thrombocytopenic patients and allows a better understanding of MK/platelet biology. Conventional mouse and human embryonic stem cells (ESCs) can be typically derived by in vitro culture of blastocysts (Martin, 1981 Thomson et al., 1998), and induced pluripotent stem cells (iPSCs) can be generated by reprogramming somatic cells using defined genetic transduction methods (Takahashi et al., 2007 Takahashi and Yamanaka, 2006 Yu et al., 2007). Megakaryocytes (MKs) are rare hematopoietic cells in the adult bone marrow and produce platelets that are critical to vascular hemostasis and wound healing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
